Development of targeted delivery systems to treat atherosclerosis
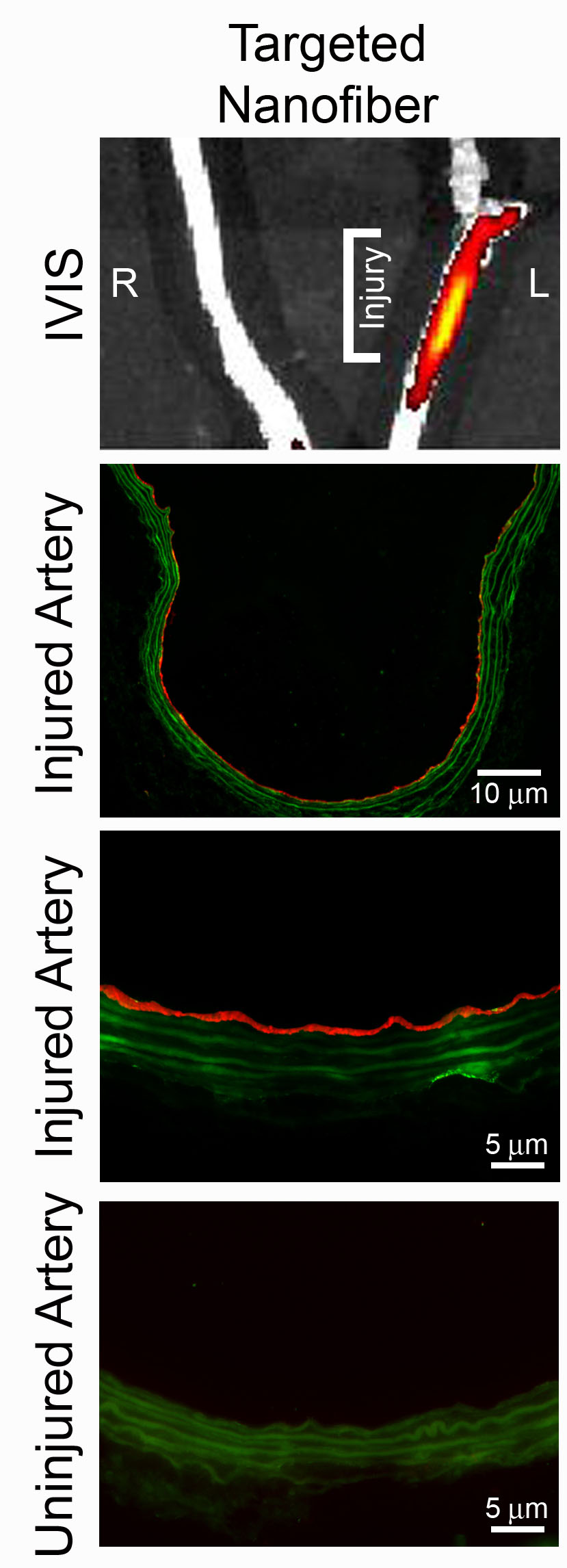
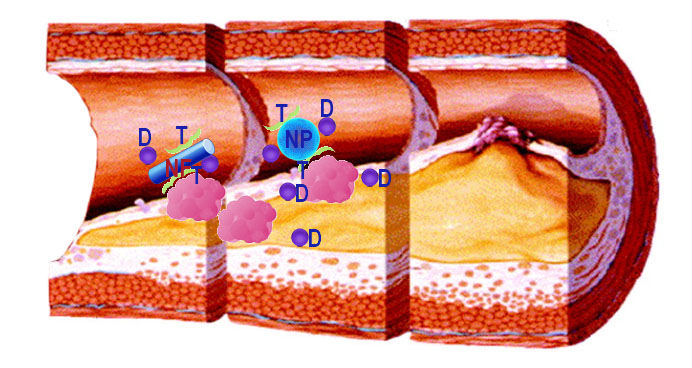
Bionanotechnology over the past decade has generated a number of potential biomedical platforms for drug delivery and diagnostics. Our lab aims to develop highly innovative targeted therapeutics to treat arterial disease. We will develop biocompatible peptide-based nanocarriers that will be delivered systemically, will target the injured or dysfuctional arterial segment, and deliver therapeutic agents to that site. We have shown that using engineered nanomaterials, it is possible to target sites of arterial injury and overcome the issues with systemic administration of redox therapies.
Our goal is to identify moieties expressed in the plaque milieu will be targeted by these nanomaterials, which in turn will direct a redox-based therapy to the site of interest. Candidates for targeting include: i) membrane proteins present in the dysfunctional endothelium such as VCAM and ICAM; and, ii) proteins expressed in the inflammatory cells recruited to the site such as AGE receptor (RAGE), folate receptor, and CD36.
An innovative and promising platform to generate targeted nanocarriers are autologous cells. Using cells of the immune system like macrophages offers the advantage that they target sites of inflammation really well. Moreover, their phagocytic capacity makes them ideal to take-up nanoparticles loaded with a therapeutic agent and deliver the payload at the site of vascular injury. Through our collaboration with Dr. Elena Batrakova, we are developing a system to deliver nrf2 activators to the injured vasculature to target arterial inflammation using macrophages as the delivery vehicle. This project is funded by the National Institute o Health.